Fourier Transform Infrared Spectroscopy (FTIR) Analysis Laboratory
FTIR Analysis is used to analyze organic materials. Bulk and small particle materials can be analyzed. FTIR microscopy analysis allows for the identification of particle as small as 10 µm. Determination of unknown materials is facilitated by spectral matching to a library of over 250,000 reference spectra.
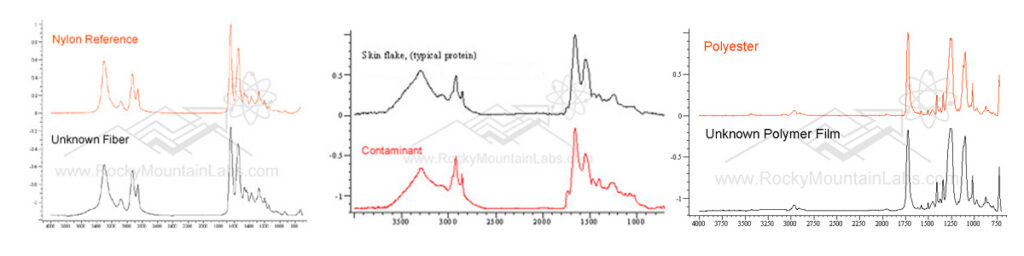
Fourier Transform Infrared Spectroscopy (FTIR) Analysis
Infrared spectroscopy is the study of interactions between matter and electromagnetic radiation. Atoms in molecules and crystals continuously vibrate with natural frequencies in the range of 1013 to 1014 cycles per second, which is the frequency of infrared radiation. Vibrations which are accompanied by a change in dipole moment cause absorption of infrared radiation. Several vibration modes may occur for a particular atomic group, each at a particular frequency which is normally independent of the other modes. If the amount of radiation absorbed by a substance is plotted against the incident wavelength, the resulting graph reflects the presence of specific chemical bonds and can therefore be used for structural identification. Whereas spectra associated with atoms are caused by electrons moving from one electronic energy level to another, inter-atomic spectra are usually characterized by either bond stretching or bending vibration modes. In addition, to these fundamental absorption bands, there are multiples of the fundamental frequencies (overtones) and frequencies which are the sum and difference of two or more fundamental frequencies, called combination lines. Wavelength is conventionally expressed in microns (µ) and frequency in wavenumber – the number of cycles per centimeter, with units of cm-1.
Infrared spectroscopy is probably the most powerful single technique available to qualitatively identify organic materials and to determine molecular structure. Mass spectrometry gives the molecular weight and formula, and nuclear magnetic resonance the number and type of protons, but only infrared indicates in a direct manner the presence of key functional groups. A match between the infrared spectra of an unknown sample and a reference sample is a simple, and almost positive, method of identification. Currently, over 130,000 reference spectra are available, most of which are available in indexed form. Spectroscopic theory will be explained in terms of very simple molecules, and then extensions will be briefly indicated to use with construction composites.
from: Composite Construction Materials Handbook, Nicholls, R. Prentice-Hall, Inc., Englewood Cliffs, NJ, 1976, pp. 528
FTIR Spectrum Analysis: Currently we match with a library of 250,000 spectra.
What is Fourier Transform Infrared Spectroscopy (FTIR)?
FTIR stands for Fourier Transform Infrared Spectroscopy. It is a type of analytical technique used in chemistry and other fields to identify and quantify the molecular components of a sample based on their infrared spectra. Infrared radiation is passed through a sample, and the molecules in the sample absorb some of the radiation at specific wavelengths, which correspond to the vibrational energies of the chemical bonds in the molecule. This absorption pattern is unique for each molecule, and FTIR can be used to identify the presence of specific functional groups in a sample, as well as to determine the purity or composition of a substance. The technique is widely used in analytical chemistry, materials science, and other fields.
How does Fourier Transform Infrared Spectroscopy (FTIR) work?
FTIR works by measuring the absorption of infrared radiation by a sample at different frequencies. The basic setup for an FTIR spectrometer consists of a source of infrared radiation, an interferometer to modulate the incoming radiation, a sample holder, and a detector.
First, the infrared radiation is generated by a source, which can be either a blackbody radiator or a laser. This radiation is then directed towards an interferometer, which splits the radiation into two beams, one of which passes through the sample and the other serves as a reference beam.
The two beams are recombined after passing through the sample and the interferometer, and the resulting interference pattern is measured by the detector. By Fourier transforming the interference pattern, the spectrum of the absorbed radiation is obtained, which is a plot of the absorption of the sample as a function of the frequency of the infrared radiation.
The resulting infrared spectrum is unique for each compound, allowing for identification and quantification of the molecular components of a sample. FTIR can be used to detect the presence of functional groups such as carbonyl, hydroxyl, and amine groups, and can be used to determine the purity and composition of a substance.
Discover Precision with Our FTIR Analysis Laboratory Services
Are you seeking accurate and reliable FTIR analysis for your research, quality control, or product development needs? Look no further! Our FTIR Analysis Laboratory is your trusted partner in the world of Fourier-Transform Infrared (FTIR) spectroscopy.
Why Choose Us?
- Cutting-Edge Technology: Our state-of-the-art FTIR Analysis Laboratory is equipped with the latest advancements in analytical instrumentation, ensuring precise and comprehensive results for a wide range of sample types.
- Expert Team: Our dedicated team of experienced scientists and analysts has a deep understanding of FTIR spectroscopy, enabling us to provide you with expert guidance, insightful data interpretation, and tailored solutions.
- Diverse Applications: We cater to a broad spectrum of industries, including pharmaceuticals, chemicals, polymers, materials science, and more. Whether you need compound identification, material characterization, or quality control, we have you covered.
- Customized Solutions: We understand that each project is unique. That’s why we offer customizable FTIR analysis solutions to meet your specific requirements, ensuring you get the data and insights you need.
- Quick Turnaround: We prioritize efficiency without compromising accuracy. Count on us for timely results, helping you make informed decisions faster.
- Quality Assurance: We adhere to rigorous quality control standards and follow industry best practices, ensuring the reliability and reproducibility of our FTIR analysis results.
- Competitive Pricing: We offer cost-effective solutions without compromising on the quality of service. Our pricing is designed to accommodate projects of all sizes.
- Confidentiality: Your data and intellectual property are safe with us. We maintain strict confidentiality and data security protocols.
- Collaborative Approach: We view our clients as partners. We’re here to collaborate with you, understand your objectives, and provide insights that drive your projects forward.
- Training and Support: Need assistance with sample preparation or data interpretation? We provide training and support services to empower you with the knowledge and skills you need.
FTIR Spectrum Analysis Services
We offer a comprehensive suite of FTIR Spectrum Analysis services designed to unravel the mysteries hidden within your samples. Our advanced Fourier-Transform Infrared (FTIR) spectroscopy capabilities, coupled with the expertise of our dedicated team, empower you to gain unprecedented insights into the chemical composition, molecular structure, and functional groups present in your materials.
Our FTIR Spectrum Analysis Services:
- Compound Identification: Discover the precise identity of organic and inorganic compounds within your samples. Our analysis pinpoints characteristic peaks and spectral patterns, enabling accurate compound identification.
- Material Characterization: Gain a deep understanding of the physical and chemical properties of your materials. We analyze spectra to elucidate material composition, crystallinity, thermal properties, and more.
- Functional Group Analysis: Identify and quantify functional groups within your samples. This service is invaluable for understanding the chemical nature of your materials.
- Quality Control: Ensure product quality and consistency with our FTIR Spectrum Analysis. We help you maintain the highest standards by analyzing samples for impurities, contaminants, and deviations from specifications.
- Polymers and Polymer Blends: Characterize polymers and polymer blends with precision. Our analysis covers molecular weight, degree of crystallinity, cross-linking, and polymer structure.
- Thin Films and Coatings: Assess the composition and thickness of thin films and coatings for applications in electronics, optics, and materials science.
- Research and Development: Collaborate with us on research projects that require FTIR spectroscopy. We offer tailored solutions, method development, and data interpretation.
- Expert Consultation: Our team of experienced scientists is always ready to provide guidance and insights into your FTIR analysis projects. We’re here to help you make the most of your results.
Fourier Transform Infrared Spectroscopy (FTIR) Testing Process
- Sample preparation: The sample must be prepared in a suitable form for analysis. This may involve grinding the sample to a fine powder, diluting it in a suitable solvent, or simply placing a solid sample on a suitable substrate.
- Measurement of the background: Before analyzing the sample, the background spectrum of the instrument must be measured. This is done by measuring the spectrum of a blank sample, such as air or a clean substrate.
- Measurement of the sample: The sample is then placed in the sample holder, and the infrared radiation is passed through it. The resulting spectrum is recorded by the detector.
- Data processing: The recorded spectrum is then processed using Fourier transform algorithms to obtain the final infrared spectrum. The spectrum is typically displayed as a plot of absorbance versus wavenumber or frequency.
- Interpretation of the spectrum: The resulting spectrum can be interpreted to identify the molecular components of the sample, including the presence of functional groups and the identity of specific compounds.
- Quantitative analysis: In some cases, the FTIR spectrum can be used for quantitative analysis to determine the amount or concentration of specific compounds in the sample.
Fourier Transform Infrared Spectroscopy (FTIR) Sampling
FTIR sampling refers to the various methods used to prepare samples for analysis by Fourier transform infrared spectroscopy (FTIR). The choice of sampling method depends on the nature of the sample and the information required.
There are several common methods of FTIR sampling, including:
- Transmission: In transmission sampling, the sample is placed between two transparent windows and the infrared radiation is passed through the sample. This method is commonly used for solid or liquid samples that are transparent to infrared radiation.
- Attenuated total reflectance (ATR): In ATR sampling, the sample is placed on a crystal with a high refractive index, such as diamond or zinc selenide, which is in contact with the infrared radiation source. The infrared radiation is absorbed by the sample at the interface between the crystal and the sample, allowing analysis of samples that are not transparent to infrared radiation, such as powders or solids.
- Diffuse reflection: In diffuse reflection sampling, the infrared radiation is reflected off the surface of the sample at a wide range of angles. This method is commonly used for analysis of samples that are not uniform or have a rough surface.
- Gas-phase sampling: In gas-phase sampling, the sample is introduced into the infrared beam in the form of a gas or vapor. This method is commonly used for analysis of gases or vapors, such as air pollutants or volatile organic compounds.
- Microscopy: FTIR microscopy involves analyzing a small area of a sample using an infrared microscope, which can provide spatially resolved infrared spectra. This method is commonly used for analysis of small particles, fibers, or biological tissues.
Fourier Transform Infrared Spectroscopy (FTIR) Techniques
Fourier transform infrared spectroscopy (FTIR) techniques are variations of the basic FTIR method that have been developed to address specific analytical challenges or to provide additional information about the sample. Some common FTIR techniques are:
- Attenuated Total Reflectance (ATR): A method in which the sample is in direct contact with a high refractive index crystal, and the infrared radiation is absorbed by the sample at the interface between the crystal and the sample. ATR is a surface-sensitive technique that is commonly used for analysis of solids, powders, and liquids.
- Reflection Absorption Spectroscopy (RAS): A technique that measures the reflection of the infrared radiation off the surface of the sample, providing information about the surface chemistry of a material. RAS is commonly used for analysis of thin films, coatings, and surfaces.
- Transmission Spectroscopy: A method in which the infrared radiation is transmitted through the sample, providing information about the molecular structure and composition of a material. Transmission spectroscopy is commonly used for analysis of liquids, gases, and transparent solids.
- Fourier Transform Raman Spectroscopy: A method that uses Raman scattering instead of absorption to provide information about the molecular vibrations of a sample. Raman spectroscopy can be used for analysis of solids, liquids, and gases, and is particularly useful for analysis of samples that do not absorb infrared radiation, such as metals.
- Two-Dimensional (2D) FTIR Spectroscopy: A method that provides information about the correlation between different vibrational modes in a sample. 2D FTIR is a powerful analytical technique that can provide information about complex chemical reactions and dynamics.
Fourier Transform Infrared Spectroscopy (FTIR) Implementation
Fourier Transform Infrared Spectroscopy (FTIR) is implemented using specialized equipment called an FTIR spectrometer. The basic components of an FTIR spectrometer include:
- Infrared source: An infrared source, typically a globar or a heated filament, provides the infrared radiation that is directed towards the sample.
- Sample compartment: The sample compartment holds the sample and provides a means for controlling the temperature, pressure, and environment of the sample.
- Interferometer: The interferometer is the heart of the FTIR spectrometer and consists of a beam splitter that divides the incoming infrared radiation into two beams, which are then directed to two mirrors. The mirrors reflect the beams back to the beam splitter, where they are recombined. The recombined beam is then directed towards the detector.
- Detector: The detector measures the intensity of the recombined beam and converts it into an electrical signal.
- Computer and software: A computer and specialized software are used to control the spectrometer and collect and analyze the data.
During FTIR analysis, the sample is placed in the sample compartment and the infrared radiation is directed towards it. The sample absorbs some of the infrared radiation, and the remaining radiation is directed towards the detector. The interferometer splits the remaining radiation into two beams, and the detector measures the intensity of the recombined beam.
The resulting signal is a complex interferogram, which must be processed using a Fourier transform to obtain the final FTIR spectrum. The FTIR spectrum analysis provides information about the molecular vibrations and chemical composition of the sample.
The implementation of Fourier Transform Infrared Spectroscopy Analysis can vary depending on the specific requirements of the analysis and the nature of the sample. Specialized accessories such as ATR or RAS cells, gas cells, or transmission cells may be used to optimize the measurement conditions and obtain the desired data.
Principles of Fourier Transform Infrared Spectroscopy (FTIR)
Fourier Transform Infrared Spectroscopy (FTIR) analysis is based on the principle that molecules absorb specific frequencies of infrared radiation corresponding to the vibrational modes of the molecular bonds. When infrared radiation is directed towards a sample, the molecules in the sample absorb some of the radiation, causing the bond to vibrate. The energy absorbed corresponds to the frequency of the bond vibration, which is characteristic of the molecular structure and composition of the sample.
The FTIR spectrometer uses an interferometer to measure the absorption of infrared radiation by the sample. The interferometer splits the incoming infrared radiation into two beams, which are then reflected back and recombined to produce an interferogram. The interferogram is a complex signal that contains information about the absorption of different frequencies of infrared radiation by the sample.
The interferogram is then subjected to a mathematical process called the Fourier transform, which converts the signal from the time domain to the frequency domain. The resulting FTIR spectrum provides a plot of the intensity of the absorbed radiation as a function of frequency. The peaks in the FTIR spectrum correspond to the vibrational modes of the molecular bonds in the sample.
FTIR spectroscopy analysis can provide a wealth of information about the molecular structure and composition of a sample. The position and intensity of the absorption peaks can be used to identify functional groups, determine the chemical composition of a sample, and detect impurities or contaminants. The intensity of the absorption peaks can also provide information about the concentration of the different components in a mixture.
The principles of FTIR spectroscopy are based on the fundamental physics of molecular vibrations and the measurement of the absorption of infrared radiation by a sample. The mathematical processing of the interferogram using the Fourier transform allows for the extraction of a wealth of information about the sample, making FTIR a valuable analytical tool in many different fields.
What is Fourier Transform Infrared Spectroscopy (FTIR) used for?
FTIR analysis is used in a wide range of applications in various fields, including chemistry, materials science and biology. Some of the common uses of FTIR include:
- Identification of unknown compounds: FTIR analysis can be used to identify unknown compounds by comparing the infrared spectra of the unknown sample with those of known compounds in a database.
- Quantification of components in a mixture: FTIR analysis can be used to determine the composition and purity of a mixture by measuring the absorbance of the sample at specific frequencies.
- Characterization of polymers: FTIR analysis is widely used in the characterization of polymers, including identification of polymer types, quantification of additives, and monitoring of polymerization reactions.
- Quality control of pharmaceuticals: FTIR analysis is commonly used in the pharmaceutical industry to ensure the quality and consistency of drugs and drug products.
List of Industries which need Fourier Transform Infrared Spectroscopy (FTIR)
Here are some industries that commonly use Fourier Transform Infrared Spectroscopy (FTIR) for various applications:
- Chemical industry: Fourier Transform Infrared Spectroscopy Analysis is used to analyze chemical reactions, identify functional groups, and monitor the composition of complex mixtures.
- Polymer industry: Fourier Transform Infrared Spectroscopy Analysis is used to analyze polymers, monitor chemical changes during processing, and characterize the chemical composition of plastics.
Strengths and Limitations of Fourier Transform Infrared Spectroscopy (FTIR)
Fourier Transform Infrared Spectroscopy (FTIR) is a powerful analytical technique with many strengths, as well as some limitations. Here are some of the strengths and limitations of FTIR:
Strengths:
- Non-destructive: FTIR analysis is a non-destructive technique, meaning that it does not damage the sample being analyzed.
- Rapid analysis: FTIR analysis can provide rapid analysis of samples, with results typically available within minutes.
- Versatility: FTIR analysis can be used to analyze a wide range of materials, including solids, liquids, and gases.
- High sensitivity: FTIR analysis can detect very small amounts of a substance, down to the parts-per-million or parts-per-billion range.
- Quantitative analysis: FTIR analysis can be used for quantitative analysis of a sample’s chemical composition, allowing for accurate determination of the amount of each component present.
- Identification of functional groups: FTIR analysis is particularly useful for identifying functional groups in organic molecules, which can be used to identify the structure of an unknown compound.
Limitations:
- Limited spatial resolution: Fourier Transform Infrared Spectroscopy Analysis has limited spatial resolution and cannot provide information on the distribution of components within a sample.
- Surface sensitivity: Fourier Transform Infrared Spectroscopy Analysis is surface-sensitive, meaning that it primarily analyzes the outermost layer of a sample. This can make it difficult to analyze samples with complex or heterogeneous surfaces.
- Sample preparation: Fourier Transform Infrared Spectroscopy Analysis requires samples to be prepared as thin films or pellets, which can be time-consuming and may alter the sample’s properties.
- Sensitivity to water: Fourier Transform Infrared Spectroscopy Analysis is sensitive to water and can be affected by moisture in the sample or the environment.
- Interference from other substances: Fourier Transform Infrared Spectroscopy Analysis can be affected by the presence of other substances in the sample, which can interfere with the analysis and lead to inaccurate results.
What is the difference between IR and FTIR?
IR and FTIR are both techniques used for infrared spectroscopy, which is a method of analyzing the interaction of molecules with infrared radiation. The main difference between IR and FTIR is in the method used to detect and analyze the infrared radiation.
IR spectroscopy typically uses a dispersive instrument to separate the different wavelengths of infrared radiation, similar to how a prism separates visible light into its component colors. The sample is exposed to a beam of infrared radiation, and the resulting spectrum is measured by detecting the intensity of each wavelength of radiation that has passed through the sample.
FTIR spectroscopy, on the other hand, uses a Fourier transform to convert the time-domain signal into a frequency-domain signal, allowing for the measurement of all wavelengths simultaneously. The sample is exposed to a beam of infrared radiation, and the resulting spectrum is measured using an interferometer, which creates an interferogram that is then processed by a computer to generate the final spectrum.
The main advantage of FTIR over IR spectroscopy is that it can provide a much higher signal-to-noise ratio, which makes it more sensitive and capable of detecting smaller amounts of sample. FTIR also provides a faster analysis time and better resolution compared to traditional IR spectroscopy.



